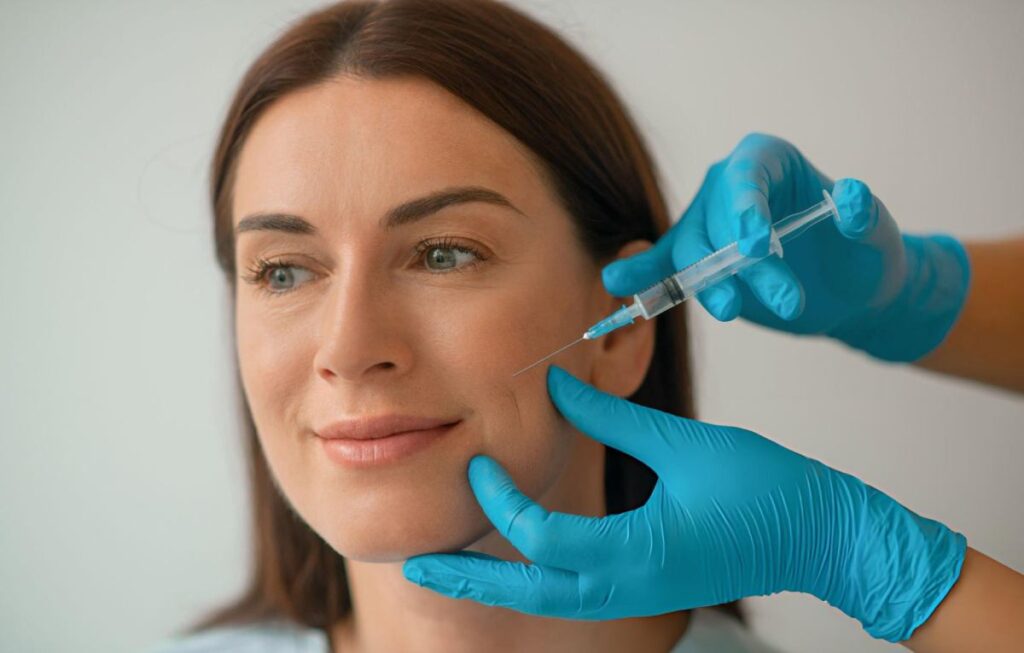
A Botox Competitor: The rejuvenating agent Azzalure is considered Botox’s primary competitor. Azzalure was previously used extensively under the brand name Dysport; however, its use was limited to the treatment of severe conditions, such as cerebral palsy, facial spasms, or strokes. Currently, this tool is actively employed by plastic surgeons.
It contains a special protein known as botulinum toxin, which is produced by the bacterium *Clostridium botulinum*. In massive doses, this protein causes the severe disease known as botulism; however, in controlled applications, it exerts a marked cosmetic effect.
A series of injections in the forehead area causes tissue immobilization, which smooths out wrinkles. However, this is not Azzalure’s primary advantage over Botox. The fact is that this new product is significantly more affordable than the “miracle gel” so popular among celebrities and high-society ladies seeking to combat the signs of aging.
For instance, in clinics across the United Kingdom, a rejuvenation treatment with Azzalure costs just £1 per unit, whereas Botox commands a price of between £4 and £6. Depending on the depth and severity of the expression lines, some cases may require the injection of up to 30 units of the drug per area.
Pharmacokinetic characteristics of the preparation
Pharmacological action takes place at the vaccination site. Presynaptic reuptake and retrograde axonal transport from the injection site are negligible. The duration of the clinical effect is 4 to 6 months. The restoration of neuromuscular activity occurs through the development of new axonal processes, which form new, functionally active neuromuscular synapses, ultimately leading to the re-establishment of muscle contractions.
When administered at therapeutic doses, Relatox does not cross the blood-brain barrier nor does it induce significant systemic effects. It is excreted via the renal route in the form of non-toxic metabolites. In 1–5% of patients, antibodies against the botulinum toxin type A-hemagglutinin complex are formed following repeated injections.
Antibody formation is favored by the administration of the drug at high doses and by the repeated injection of small doses at short intervals. Should antibodies against botulinum toxin type A be formed, the efficacy of the subsequent response may be reduced.
Side effects
- Local reactions: 2–5% of cases — microhematomas (up to 7 days), pain at the injection site (up to 1 day).
- Systemic reactions: occur when a high dose is used (more than 200 units) — mild general weakness lasting up to one week.
- Reactions associated with diffusion into tissue groups located near the needle insertion point, depending on the area of administration of botulinum toxin type A. In the treatment of blepharospasm and hemifacial spasm: ptosis (5–10%), lacrimation (0.5–1%); rarely: ectropion, keratitis, diplopia, entropion, ecchymosis. When injected into both sternocleidomastoid muscles: dysphagia (2–5%). Generally, these side effects do not require additional therapy and resolve within one month following the injection.